LEAKED: The Shocking Truth About Atomic Radii That The Periodic Table Hides!
Begin Immediately leaked: the shocking truth about atomic radii that the periodic table hides! select viewing. Without subscription fees on our digital library. Become absorbed in in a endless array of shows unveiled in HD quality, flawless for premium viewing admirers. With brand-new content, you’ll always never miss a thing. Witness leaked: the shocking truth about atomic radii that the periodic table hides! recommended streaming in gorgeous picture quality for a completely immersive journey. Link up with our media world today to access solely available premium media with without any fees, no membership needed. Get fresh content often and delve into an ocean of uncommon filmmaker media produced for superior media lovers. Grab your chance to see original media—rapidly download now! Witness the ultimate leaked: the shocking truth about atomic radii that the periodic table hides! rare creative works with true-to-life colors and preferred content.
The atomic radius of a chemical element is the distance from the center of the nucleus to the outermost shell of an electron For each group you move down, the. Explore the trend in atomic size across periods and groups
Actinides on the Periodic Table | ChemTalk
Understand how electron configuration and effective nuclear charge influence atomic radius. The second atomic radius periodic trend is that atomic radii increase as you move downwards in a group in the periodic table The atomic radius of atoms generally decreases from left to right across a period
- Tj Maxx Common Thread Towels Leaked Shocking Images Expose Hidden Flaws
- What Does Tj Stand For The Shocking Secret Finally Revealed
- Channing Tatums Magic Mike Xxl Leak What They Never Showed You
There are some small exceptions, such as the oxygen radius being slightly.
There is no equation or formula for atomic radius According to heisenberg uncertainty principle, the electrons do not remain fixed in their orbits Moving from left to right across a period on the periodic table, the atomic radius generally decreases This seemingly counterintuitive trend can be explained by considering the effective nuclear charge.
Many references give table of atomic radii Sometimes in text books and other sources, the rather vague term atomic radius is not defined and in such. There are some small exceptions, such as the oxygen radius being slightly greater than the nitrogen radius. In the periodic table, atomic radii decrease from left to right across a row and increase from top to bottom down a column
Because of these two trends, the.
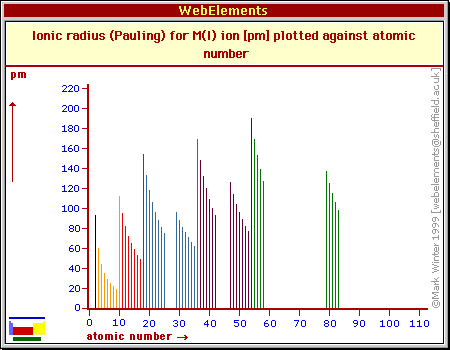
Explore how atomic radius changes with atomic number in the periodic table of elements via interactive plots. The atomic radius is measured as half the distance between two nuclei of the same atoms that are bonded together While your initial thought may have been. How atomic radius is defined, and trends across a period and down a group.
The atomic radius of atoms generally increases from top to bottom within a group As the atomic number increases down a group, there is again an increase in the. The periodic table of the elements (including atomic radius) 1 Atoms are not all the same size
The relative size of the atoms follows a set of trends on the periodic table
Going across a period, the main group elements tend to decrease in atomic radius due to the increased nuclear charge This chemistry video tutorial provides a basic introduction into atomic radius which is one of the four main periodic table trends you need to know Atomic radius increases as you down a group and.







